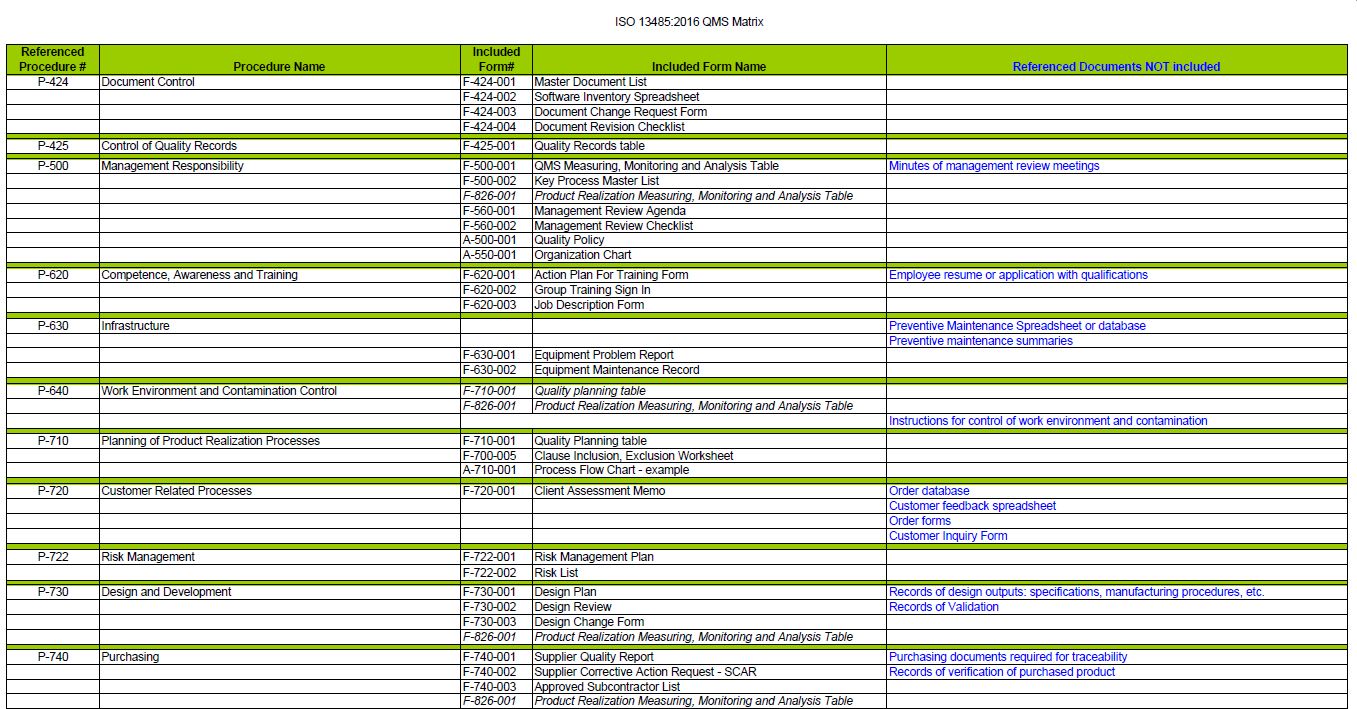 
To be useful, audits must be performed and presented in a meaningful approach. A “value-add” perspective for audits is needed!Īuditing is a cost-effective means of improving the organization. The emphasis of ISO 13485:2016 focuses on process effectiveness, efficacy, and safety and thus requires a different approach to auditing. Students gain the necessary auditing skills through a balance of formal classroom tutorials, practical role-playing, group workshops, and open forum discussions. Experienced instructors explain the clauses of ISO 13485:2016 in detail and guide students through internal audits to assure value to their business and regulatory authorities. In addition, this class will teach the principles and practices of effective internal audits in accordance with ISO 19011:2018, “Guidelines on Auditing Management Systems”. QSG’s ISO 13485:2016 Internal Auditor workshop teaches the fundamental concepts of ISO 13485:2016 and will explain in detail how to integrate the requirements into a quality management system. At the heart of ISO 13485:2016 is a comprehensive risk management process meant to be implemented in all medical device-related quality management processes within an organization, including processes that occur after the medical devices are placed on the market. It embodies an agreed upon, repeatable way of managing design, production, validation, quality, and risk management. ISO 13485:2016 is the quality management system that governs medical devices upon which compliance with regulatory and customer compliance can be built. MA Workforce Training Fund Express Grant.MA Workforce Training Fund General Grant.Outsourced PPAP Review & Disposition (APQP Processes). 
Auditing Core Tools & Customer-Specific Requirements.Quality / Regulatory Management Systems.EU’s In Vitro Diagnostic Medical Devices Regulation (IVDR).Cybersecurity Maturity Model Certification (CMMC).Process Improvement Tools / Technical Skills.Quality Management & Quality Control Support Services.National Aerospace and Defense Contractors Accreditation Program (NADCAP).Management System Standards – Implementation & Integration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
